Learning Objectives
Understand the pathophysiology of Methemoglobinemia. Master the biochemical shift from ferrous (Fe2+) to ferric (Fe3+) iron, identify common inducing agents, and recognize the classic clinical presentation and treatment for the USMLE Step 1.
1. The Biochemical Shift: Fe2+ vs. Fe3+
Normal hemoglobin contains iron in the reduced ferrous (Fe2+) state, which is capable of binding oxygen. In methemoglobin, iron is oxidized to the ferric (Fe3+) state.
| State | Mnemonic | O2 Binding Ability |
|---|---|---|
| Ferrous (Fe2+) | “Just the 2 of us” (Fe and O2) | Normal; binds O2 effectively. |
| Ferric (Fe3+) | “Ferric is 3-freakish” | Reduced; does not bind O2. |
The “Left Shift” Trap: Not only does the Fe3+ subunit fail to bind oxygen, but it also increases the affinity of the remaining Fe2+ subunits for oxygen. This prevents oxygen from being released to the tissues, causing tissue hypoxia.
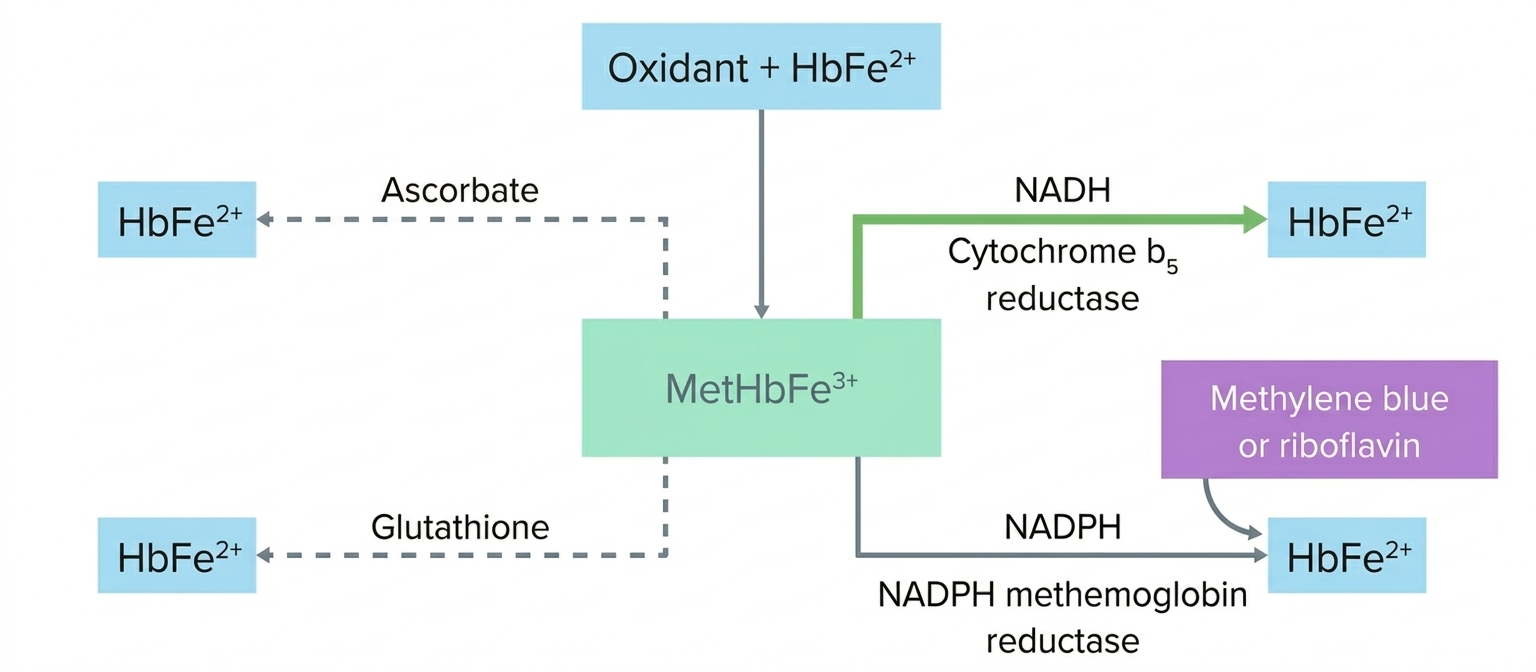
2. Clinical Presentation and Causes
Methemoglobinemia is a clinical emergency often triggered by specific medications or environmental exposures.
| Category | Details |
|---|---|
| Common Inducers | Dapsone, Local anesthetics (Benzocaine), Nitrites (polluted water/well water), Sulfonamides. |
| Physical Exam | Cyanosis (bluish skin) and Chocolate-colored blood. |
| Classic Clue | Cyanosis that does not improve with 100% supplemental oxygen. |
3. Management and Cyanide Connection
Methemoglobinemia has a unique relationship with Cyanide poisoning and specific antidotes.
| Management Type | Intervention | Mechanism |
|---|---|---|
| Acute Treatment | Methylene Blue | Acts as an electron donor to reduce Fe3+ back to Fe2+. |
| Chronic/Adjunct | Vitamin C | Reducing agent (slower than Methylene Blue). |
| Cyanide Antidote | Nitrites (to induce MetHb) | Fe3+ binds cyanide tightly, pulling it away from cytochrome c oxidase. |
Activity:
High-Yield Clinical Pearls:
- The Pulse Ox Gap: Conventional pulse oximetry is unreliable in methemoglobinemia; it often stays around 85% regardless of the true oxygen levels.
- O2 Parameters: In methemoglobinemia, PaO2 is normal (dissolved O2 is fine), but SaO2 and Total O2 content are decreased.
- Newborns: Infants are more susceptible because they have lower levels of NADH-methemoglobin reductase, the enzyme that normally keeps iron in the Fe2+ state.

You must be logged in to post a comment.