Learning Objectives
Compare the structural and functional differences between Hemoglobin and Myoglobin. Master the concepts of positive cooperativity, allosteric regulation, and the physiological roles of these oxygen-binding proteins for the USMLE Step 1.
1. Structure and Binding of Hemoglobin
Hemoglobin (Hb) is a complex protein designed for the efficient transport and unloading of oxygen throughout the body.
| Feature | Details | Function |
|---|---|---|
| Subunit Composition | Adult Hb (HbA1): 2 alpha and 2 beta subunits. | Allows for complex allosteric interactions. |
| Binding Capacity | Each tetramer binds 4 molecules of O2. | Maximizes O2 carrying capacity per RBC. |
| Buffering | The protein component binds H+ and CO2. | Crucial for maintaining blood pH. |
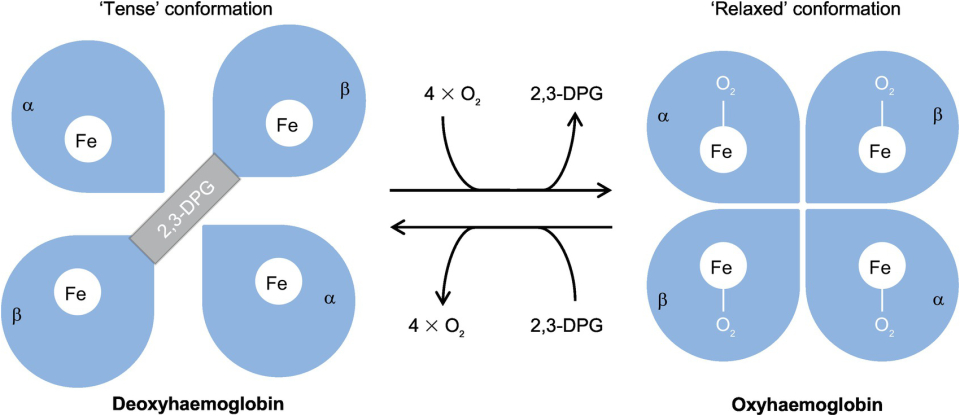
2. Allostery and Positive Cooperativity
Hemoglobin is an allosteric protein, meaning its shape (and affinity) changes depending on whether oxygen is bound.
| State | Description | Oxygen Affinity |
|---|---|---|
| Tense (T) State | Deoxygenated hemoglobin. | Low Affinity (Promotes O2 unloading at tissues). |
| Relaxed (R) State | Oxygenated hemoglobin. | High Affinity (300x higher; promotes O2 loading at lungs). |
Positive Cooperativity: The binding of one O2 molecule makes it easier for the next O2 to bind. This results in the characteristic Sigmoidal (S-shaped) dissociation curve.
3. Myoglobin: The Muscle Reservoir
Unlike hemoglobin, myoglobin is a monomeric protein found within muscle cells.
| Characteristic | Myoglobin Details | Comparison to Hb |
|---|---|---|
| Structure | Single polypeptide chain; 1 heme. | No cooperativity (Hyperbolic curve). |
| Affinity | Very high affinity for O2. | Much higher than Hb; it only releases O2 at very low PO2. |
| Role | O2 storage in the heart and skeletal muscle. | Essential for aerobic metabolism during heavy exercise. |
Activity:
High-Yield Clinical Pearls:
- Fetal Hemoglobin (HbF): Composed of 2 alpha and 2 gamma subunits. It has a higher affinity for O2 than adult Hb, allowing the fetus to “steal” oxygen from maternal blood.
- The Sigmoid Advantage: Hemoglobin’s sigmoidal curve allows it to be nearly saturated in the lungs (high PO2) but release a large amount of O2 with only a small drop in PO2 at the tissues.
- Myoglobinuria: If muscle is damaged (Rhabdomyolysis), myoglobin is released into the blood and cleared by the kidneys, which can cause acute tubular necrosis.

You must be logged in to post a comment.