Learning Objectives
Understand the three primary methods of Carbon Dioxide Transport in the blood. Master the biochemical reactions within the RBC, the significance of the Chloride Shift, and the physiological importance of the Haldane Effect for the USMLE Step 1.
1. The Three Forms of 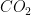 Transport
Transport
Carbon dioxide is much more soluble in blood than oxygen, but it must still be managed carefully to maintain acid-base balance. Latex
| Form | Percentage | Mechanism |
|---|---|---|
| Bicarbonate ( |
90% | |
| Carbamino Compounds | 5% | |
| Dissolved |
5% | Free |
2. The RBC Reaction and Chloride Shift
At the systemic tissues, enters the Red Blood Cell (RBC) and undergoes a critical transformation. This process involves a specific exchange to maintain electrical balance.
| Step | Reaction/Movement | Consequence |
|---|---|---|
| Conversion | ||
| Chloride Shift | Maintains electrical neutrality via the |
|
| Osmotic Effect | Water follows |
Venous RBCs swell slightly, increasing venous hematocrit. |
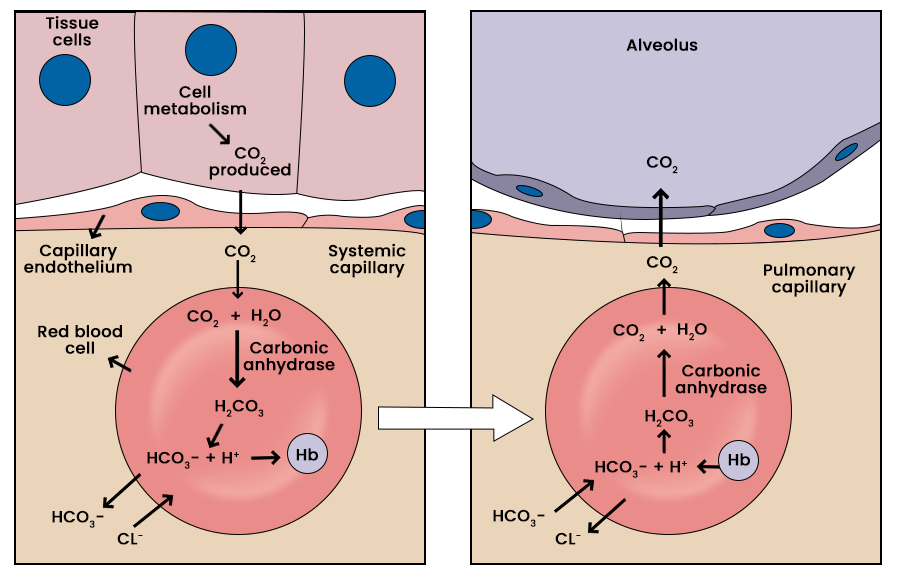
3. The Haldane Effect (In the Lungs)
The Haldane effect describes how the oxygenation status of hemoglobin affects its ability to carry .
| Location | Event | Result |
|---|---|---|
| Pulmonary Capillaries | Hemoglobin becomes more acidic and releases |
|
| Equilibrium Shift |
Activity:
High-Yield Clinical Pearls:
- Bohr vs. Haldane: The Bohr Effect (tissues) is how
kicks
off Hb. The Haldane Effect (lungs) is how
kicks
off Hb.
- Carbonic Anhydrase: This enzyme is inhibited by Acetazolamide, which interferes with
reabsorption and
transport.
- Carbaminohemoglobin:
binds to the N-terminus of globin, while
and
bind to the Iron in heme.

You must be logged in to post a comment.