Learning Objectives
Master the concept of Ion Trapping and how urine pH manipulation affects drug elimination. Understand the relationship between and pH, and identify the clinical treatments for specific drug toxicities for the USMLE Step 1.
1. The Principle of Ion Trapping
The kidney filters drugs into the renal tubule. To be excreted, a drug must remain in the tubular lumen and not be reabsorbed across the lipid membrane back into the blood.
Lipid Soluble (Neutral/Non-ionized): Can cross membranes; easily reabsorbed.
Water Soluble (Charged/Ionized): Cannot cross membranes; stays in the urine (“Trapped”) and is cleared quickly.
| Drug Type | Environment for Trapping | Clinical Examples |
|---|---|---|
| Weak Acids | Alkaline (High pH) environment ( |
Phenobarbital, Methotrexate, Aspirin (Salicylates). |
| Weak Bases | Acidic (Low pH) environment ( |
Amphetamines, Tricyclic Antidepressants (TCAs). |
2. Clinical Management of Overdose
We manipulate urine pH to change the ionization state of the drug, thereby accelerating its elimination.
| Therapeutic Goal | Treatment Agent | Mechanism |
|---|---|---|
| Alkalinize Urine | Sodium Bicarbonate | Increases pH |
| Acidify Urine | Ammonium Chloride | Decreases pH |
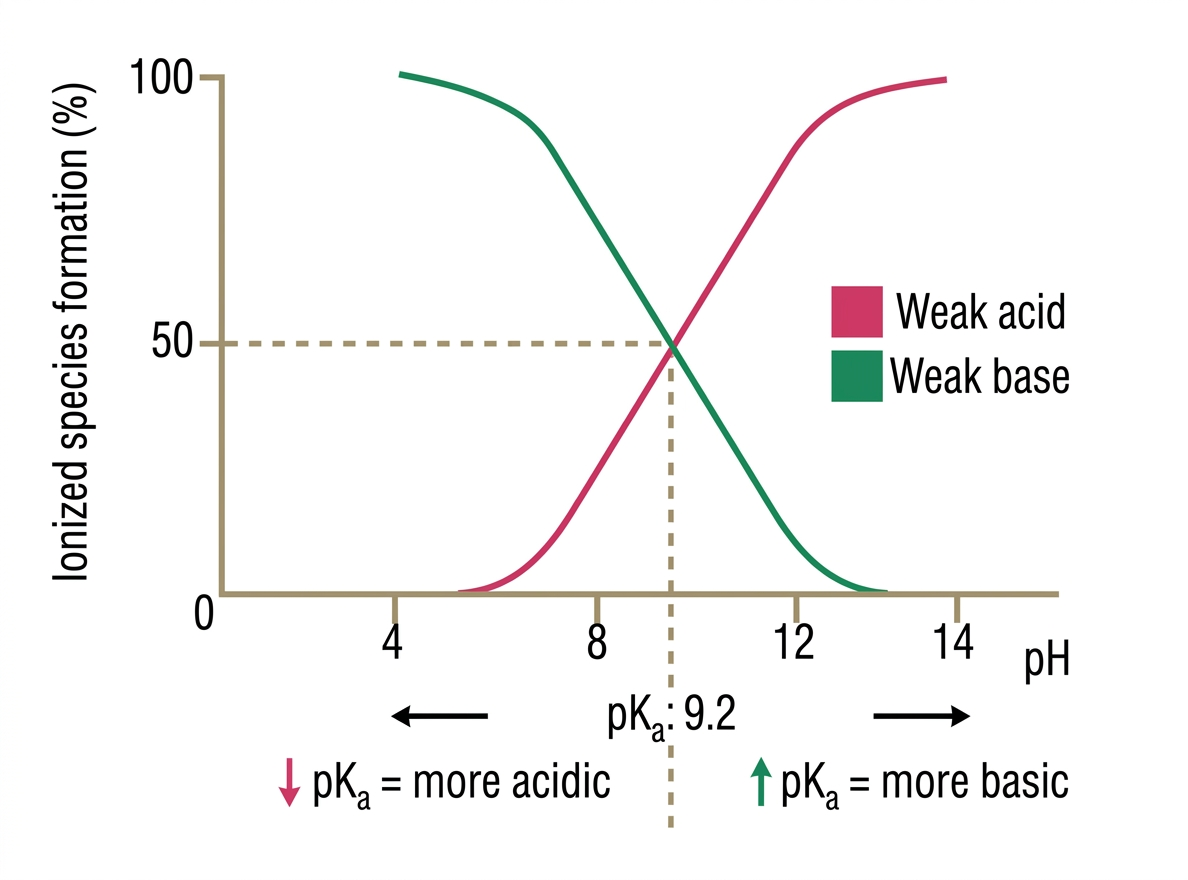
3.  and Ionization Status
and Ionization Status
The is the pH at which a drug is 50% ionized and 50% non-ionized.
| Condition | Weak Acid Status | Weak Base Status |
|---|---|---|
| Mostly Ionized (Trapped) | Mostly Neutral (Reabsorbed) | |
| Mostly Neutral (Reabsorbed) | Mostly Ionized (Trapped) |
4. High-Yield Exception: TCA Toxicity
While TCAs are weak bases, the use of Sodium Bicarbonate here is primarily for cardioprotection, not elimination.
| Feature | Clinical Detail |
|---|---|
| Primary Goal | To treat cardiac toxicity (e.g., QRS widening, arrhythmias). |
| Mechanism | Sodium load overcomes the sodium channel-blocking activity of TCAs. |
| Elimination | Does NOT accelerate drug elimination (alkaline pH keeps bases neutral). |
Activity:
High-Yield Clinical Pearls:
- Mnemonic: “Like dissolves like, opposites distract.” (Acid in acid = neutral/reabsorbed; Acid in base = ionized/trapped).
- Salicylate Duality: Aspirin overdose causes a respiratory alkalosis initially, but renal elimination is boosted by making the urine more alkaline.
- TCA Management: On the USMLE, if you see QRS widening in an overdose, the answer is almost always Sodium Bicarbonate, regardless of the drug’s
.
You must be logged in to post a comment.