Learning Objectives
Master the principles of Enzyme Kinetics. Understand the relationship between substrate concentration and reaction velocity, interpret Michaelis-Menten and Lineweaver-Burk plots, and distinguish between different types of enzyme inhibition for the USMLE Step 1.
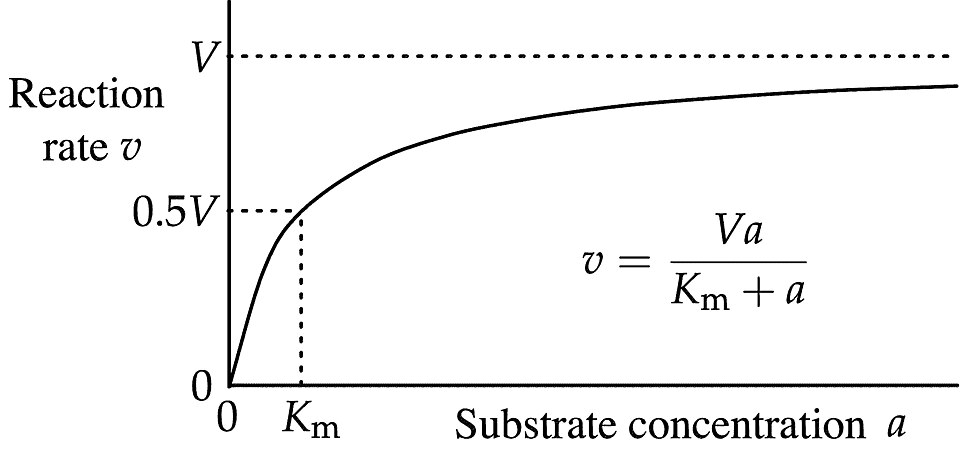
1. Michaelis-Menten Kinetics
Most enzymatic reactions follow a hyperbolic curve, describing how the reaction velocity () changes with substrate concentration (
).
| Variable | Definition & Properties |
|---|---|
| The maximum velocity of the reaction. It is directly proportional to enzyme concentration. | |
| The |
|
| Cooperativity | A sigmoidal curve (instead of hyperbolic) indicates positive cooperativity (e.g., Aspartate transcarbamoylase). |
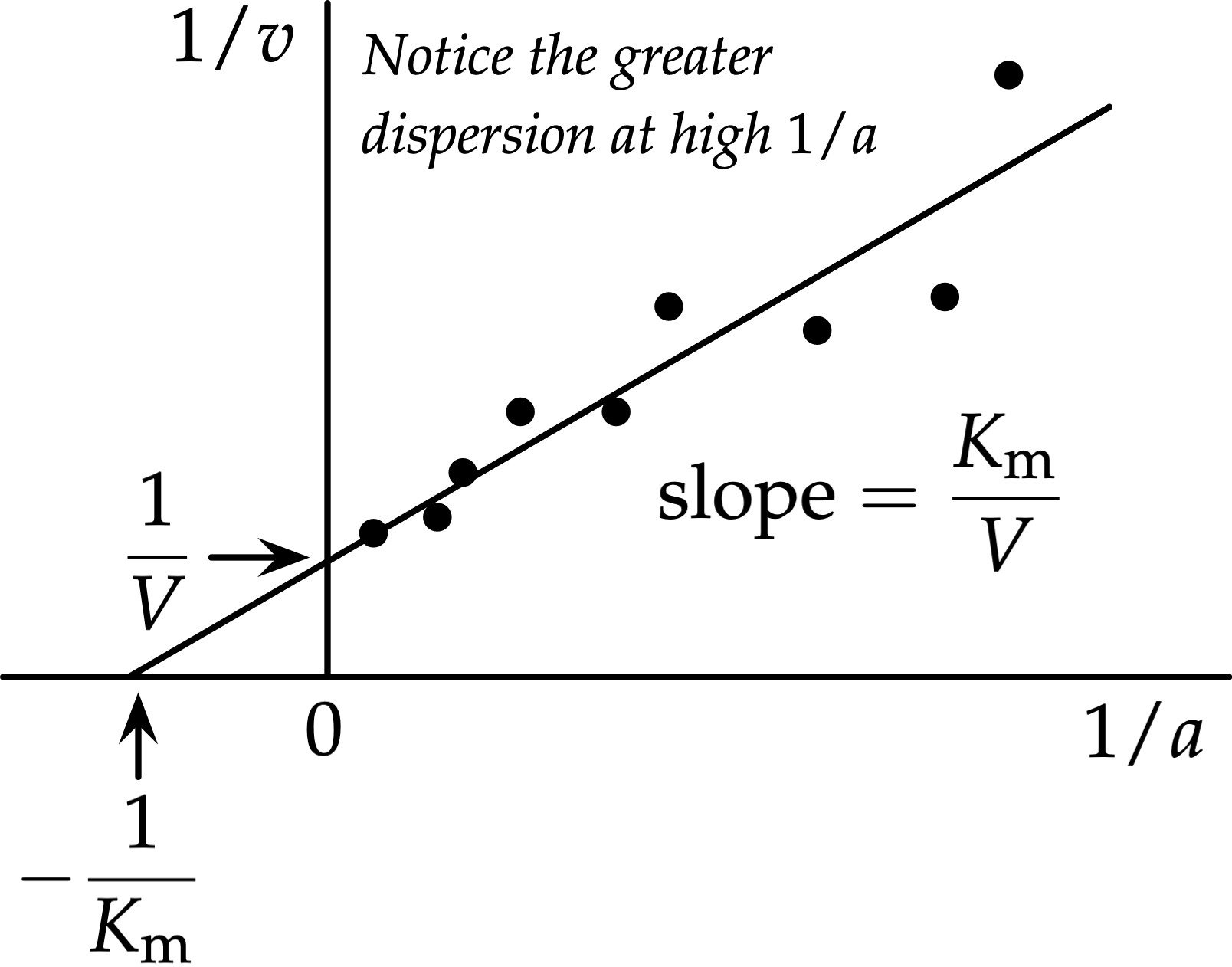
2. Lineweaver-Burk Plot
The Lineweaver-Burk plot is a double-reciprocal transformation ($1/V$ vs. $1/[S]$ ) that creates a straight line, making it easier to determine and $K_m$.
| Plot Feature | Mathematical Value | Interpretation |
|---|---|---|
| Y-intercept | The closer to 0, the higher the |
|
| X-intercept | The closer to 0, the higher the |
|
| Slope | Represents the ratio of affinity to maximum velocity. |
3. Effects of Enzyme Inhibition
Inhibitors change the kinetics of enzymatic reactions by either competing for the active site or binding elsewhere to reduce catalytic efficiency.
| Feature | Competitive (Reversible) | Noncompetitive |
|---|---|---|
| Resemble Substrate? | Yes | No |
| Bind Active Site? | Yes | No (Allosteric site) |
| Overcome by |
Yes | No |
| Effect on |
Unchanged | Decreased ( |
| Effect on |
Increased ( |
Unchanged |
| Pharmacodynamics |
Activity:
4. Irreversible Competitive Inhibition
Irreversible competitive inhibitors bind covalently to the active site. Because they cannot be displaced by substrate, they behave kinetically like noncompetitive inhibitors.
| Parameter | Change | Reasoning |
|---|---|---|
| Decreased ( |
Reduces the effective number of available enzymes. | |
| Unchanged | The remaining free enzymes have normal affinity. | |
| Pharmacodynamics | The maximal effect of the drug/enzyme is lowered. |
Activity:
High-Yield Clinical Pearls:
- Kompetitive Inhibitors: Remember that Kompetitive inhibitors increase Km. On a Lineweaver-Burk plot, the lines will cross at the Y-axis (same
).
- Noncompetitive Inhibitors: These do not cross at the Y-axis because the
is different. They look like they are “pinched” together at the X-axis (same
).
- Statins: A classic clinical example of reversible competitive inhibitors of HMG-CoA reductase.
You must be logged in to post a comment.